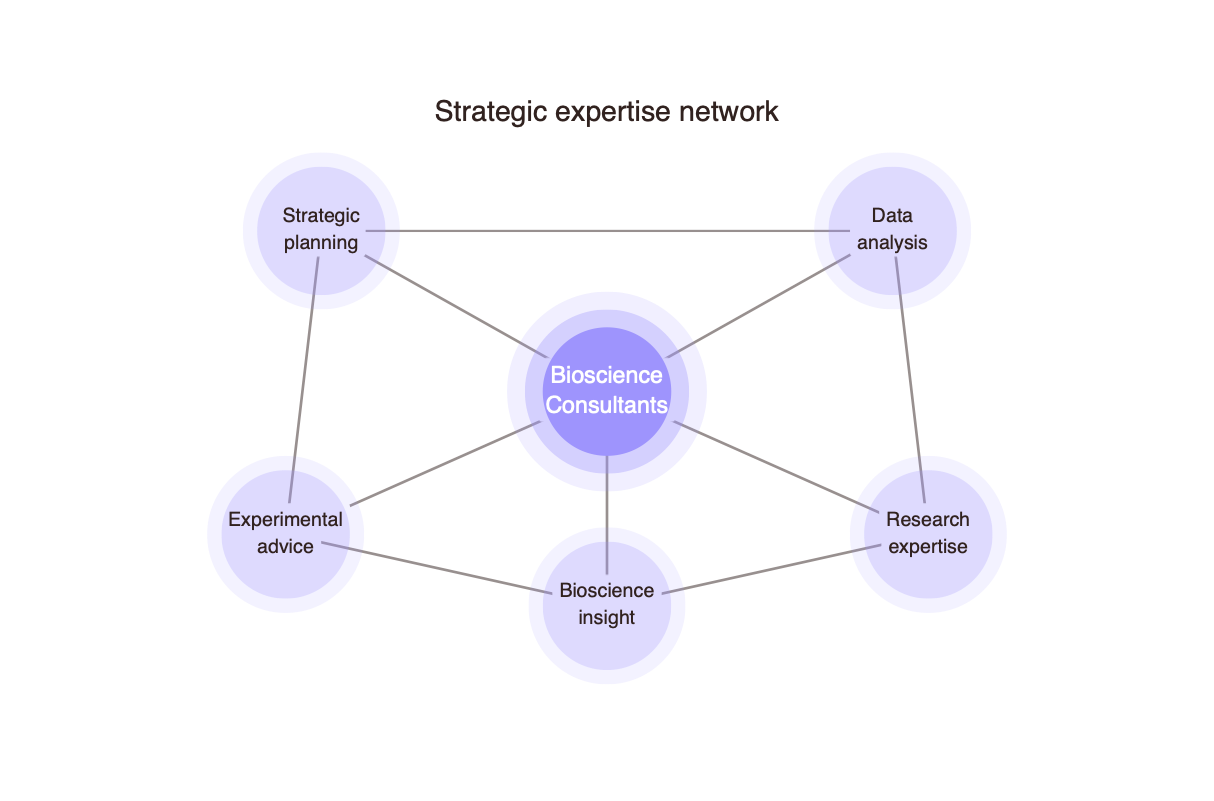
Planning
I help establish strong foundations for success by facilitating robust project and experimental design. This includes evaluating the commercial potential of concepts and ongoing projects, as well as supporting early-stage business planning. Drawing on extensive experience and a deep understanding of regulatory requirements, I ensure that plans are both scientifically sound and aligned with industry and regulatory expectations from the outset. My approach is practical and thorough, enabling informed decision-making from the outset..
Strategy
Drawing on a broad knowledge base and extensive industry experience, I work with clients to identify opportunities, build strategic partnerships, and define clear development pathways. I provide guidance across all stages of preclinical antimicrobial development and early clinical studies, helping to navigate complex scientific and regulatory landscapes while positioning projects for long-term success..
Execution
I deliver high-quality, detail-oriented support to to translate strategy into tangible outcomes. My services include advanced data analysis and the preparation of professional scientific materials such as reports, manuscripts, presentations, and regulatory documentation. With a focus on clarity, accuracy, and impact, I ensure that all outputs meet the highest academic, clinical, and commercial standards required for decision-making, communication, and compliance.
a